BY RACHEL ARNESEN
“Ebola in the air? A nightmare that could happen.”
“Ebola: World Goes on Red Alert.”
“Ebola: ‘The ISIS of Biological Agents.’”
These headlines, taken from real CNN and BBC articles, capture all too well the fear mongering that occurred during the most recent outbreak of Ebola in West Africa. From 1996 to 2009, 53% of emerging global infectious diseases had their roots in Africa, the continent that lags furthest behind in disease detection and warning.1 Ebola is an example of a zoonotic virus, or a virus that has “spilled over” from animals to humans. Zoonotic viruses, the most frequently emerging human pathogens, constitute over 65% of all pathogens discovered in humans since 1980.1 Despite the predictions of prominent virologists Macfarlane Burnet and David O. White in the 1970s that the “most likely forecast about the future of infectious disease is that it will be very dull,” the emergence of zoonotic diseases such as HIV/AIDS, Ebola, and SARS demonstrates that the field of infectious disease remains far from boring.2 The continued emergence of such zoonoses is rooted in an evolutionary past and has the potential for devastating public health outcomes. The headlines mentioned above, though alarmist, raise important questions about the danger of these zoonoses, why they emerge, and what we can do to control them.
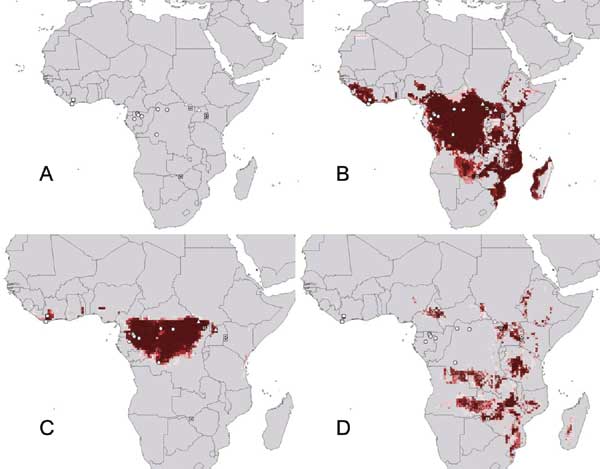
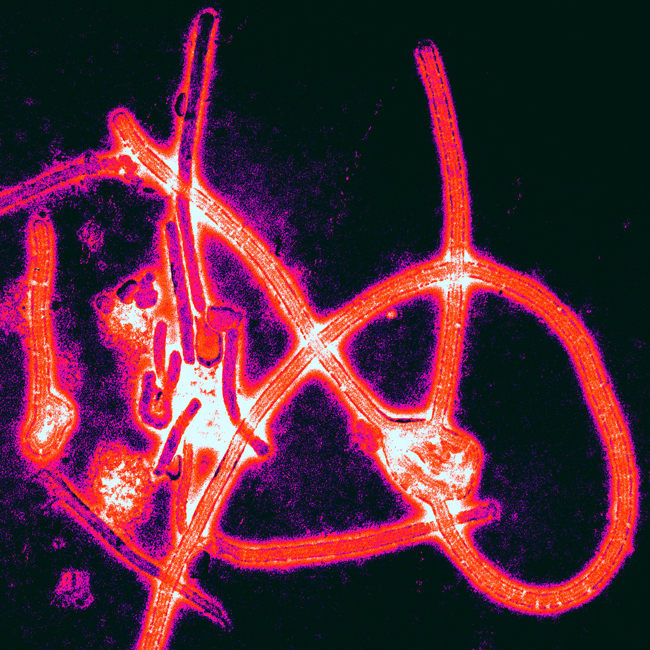
In 1976, the Ebola virus was identified in what is now the Democratic Republic of the Congo (DRC).3 Since then, subsequent outbreaks of this filovirus have occurred throughout central and western Africa, with the largest outbreak ending only recently in West Africa. As of mid-July 2015, this outbreak had caused over 27,000 cases and 11,000 deaths in Guinea, Liberia, Nigeria, and Sierra Leone.4 Marburg virus, the only other pathogen in the filovirus family, was discovered in 1967 when several laboratory workers in Marburg, Germany, fell severely ill. Scientists traced the outbreak back to the researchers’ study subjects: African green monkeys from Uganda. During that outbreak, a total of 31 cases of Marburg were detected, with seven fatalities.5 Large natural outbreaks of Marburg have occurred since, including an outbreak in the Democratic Republic of Congo in 1998, which resulted in 149 cases and 123 deaths (83% fatality rate), and a 2004 Angola epidemic that led to 252 cases and 227 deaths (90% fatality rate).5 In 2012, a novel coronavirus emerged in the Middle East, which soon earned the name Middle East respiratory syndrome (MERS). Since September 2012, the disease has caused 1,611 confirmed cases of MERS and 575 deaths across 26 countries.6 Ebola, Marburg, and MERS represent just three examples of diseases that spread from animals into humans, and then among humans as well.
Of 335 emerging infectious disease (EID) events cataloged between 1940 and 2004, 60.3% were classified as zoonoses, with 71.8% of those zoonotic events caused by pathogens of “wildlife origin.” 7 Over the course of those 65 years, pathogens originating in wild animals caused 43% of all EID.7 However, from 1990-2000, the proportion of EID caused by zoonotic pathogens originating in wild animals jumped to 52%, a large statistical increase.7 This rise begs an important question: what is driving the increase in spillover events in recent years? The accelerated emergence of zoonotic diseases calls into focus a need to understand the evolutionary processes behind spillover virulence, the phenomenon in which a disease moves from an animal host into the human population and causes illness. What are the driving forces behind these spillover events? More specifically, how have human actions increased the likelihood of zoonoses?
Filoviruses such as Ebola move from one live host to another, thus they must balance an increase in virulence, defined as the pathogen’s ability to cause disease, against transmission ability. This tradeoff between the length of time the host survives after a pathogenic invasion and the rate at which it is transmitted to others is known as the virulence-transmission tradeoff. Bats are the long-term host for filoviruses, thus filovirus infection in bats is asymptomatic and non-lethal. One reason this host-pathogen relationship may have evolved is because the metabolism and core temperature of bats increases greatly during flight.7 Since the inhospitable environment of the bat acts as a selective force on pathogens, such viruses would have to evolve to weather those changes. Given that tradeoffs are central to evolution, such an adaptation would have to be accompanied by another change: in this case, decreased pathogen virulence in the bat host. Bats and filoviruses have coevolved so that the high bat body temperature during flight does not kill the filoviruses, and conversely, the presence of filoviruses does not harm the bats. However, when the filovirus encounters a naïve host with less hostile conditions, such as the human body with its lower temperatures, the virulence level skyrockets. Within a naïve host, such as a human, the parasite proliferates as increased virulence is selected for.8 When a filovirus outbreak ends, the disease goes back into “hiding” in its natural reservoir. Since the pathogen is practically harmless to its natural host, it is near impossible to predict the next episode of spillover virulence. This difficulty of detection begs the question: what determines whether or not a disease will spill over into the human population?
Because Ebola and Marburg viruses have equilibrated to the bat immune system, they can withstand large fluctuations in body temperature, rendering the typical human fever response relatively ineffective. An individual parasite does not “think” on an evolutionary time scale, so this ability did not result from conscious decision-making. Rather, once the virus enters a naïve host, it can proliferate and grow unhindered. However, such rapid growth is not conducive to a sustained epidemic or outbreak. Diseases such as Ebola and Marburg tend to kill their hosts more quickly than they infect new ones, preventing the diseases from becoming established in the new host population.
In addition to the individual, biological characteristics of viruses that predispose them to spillover events, human actions have increased the rates and risks of zoonotic events. Human behaviors affect animal-environment interactions, human-environment interactions, and animal-human interactions. These non-biological drivers fall into two different categories: proximate drivers and ultimate drivers. Proximate drivers occur on a relatively local level, and include population movement and migration, habitat changes, and alterations in food and water usage.9 Ultimate drivers occur on a broad temporal and geographic scale, and they shape the proximate drivers. Examples of ultimate drivers include climate change and variability, changes in land use, and changes in animal interactions.9
The proximate drivers of movement/migration and habitat change primarily affect zoonosis emergence by expanding the human-wildlife interface. When habitat destruction, flooding, drought, etc., displace animals from their natural habitats, they may be forced to move to new areas, frequently located closer to human population than before.10 This convergence provides more opportunity for human-wildlife interactions and more opportunity for a disease harbored by a wild animal to jump into the human population. The same process can happen in reverse when humans migrate to new locations.10
Though a less direct mechanism than the drivers described above, climate change also drives zoonoses, affecting the rate of spillover events through four different mechanisms. First, climate change induces a “range shift” in human or animal populations, thus increasing the number of interactions between the host and the vector. Second, it changes the population density of the host or vector, shifting the frequency of interactions between the two. Third, it augments the infection prevalence, which could change how often humans come into contact with an infected animal. Lastly, climate change alters the pathogen load through “changes in rates of pathogen reproduction, replication, or development,” thus affecting the ability of the pathogen to infect a human even if contact is established.11 Ecological niche models predict the location of potential filovirus outbreaks to mainly concentrate in sub-Saharan Africa, with Ebola outbreaks occurring in the “humid rain forests of central and western Africa,” and Marburg outbreaks occurring in “the drier and more open areas of central and eastern Africa.”12 By affecting these ranges, climate change could render our current modeling techniques inaccurate and increase the risk of spillover events in previously unaffected or lightly affected areas.
Growing land use on large scales, another ultimate driver of spillover events, has likely contributed significantly to the increased rate of zoonoses in recent years. Such changes include deforestation, pasture to cropland conversions, crop intensification, and urbanization.9 These alterations expand the human-wildlife interface in ways similar to those of the proximate drivers mentioned above. As humans expand further into previously uninhabited territory, they augment the risks of new or increased contact with species that previously lived separately from them.9 The widely seen practices of deforestation and agricultural intensification encroach on land previously prowled only by wildlife. Cutting down forests where, for example, bat species live, forces the bats to find a new habitat. Since less and less land remains unexplored by the human species, these bats will now frequently share their habitat with human populations, increasing the risk of zoonotic events.
Changes in animal management and food sources can be viewed as both proximate and ultimate drivers of zoonotic disease emergence. When dealing with zoonotic diseases originating in wildlife, not domesticated animals, the hunting, trade, and consumption of bush meat play crucial roles. Any meat obtained from wild animals, a frequent source of food in many of the regions that have experienced filovirus outbreaks, is considered bush meat.13 A study conducted in 2002 in the Congo basin, a hot zone for Ebola, estimated that over 282.3 grams of bush meat is eaten per person per day, with a total of 4.5 million tons of bush meat extracted annually.13 Many zoonotic diseases, including Ebola and Marburg, can pass to human hosts when people eat or even handle infected meat. Increased demand for bush meat is likely associated with the increased incidence of zoonotic diseases.
No single “most important” factor drives filovirus spillover events. These events occur when the bat-human-environment interactions line up in a way that facilitates virulence spillover and subsequent propagation throughout a human population. This leads to an important question: What does the future zoonotic disease landscape look like for the human population?
A massive, prolonged filovirus event does not seem to be a major risk to the human population. Although the most recent outbreak of Ebola in West Africa is certainly worrisome, the epidemic appears to have ended approximately two years after it began ravaging the region. While the 27,000+ cases and 11,000+ deaths it caused make the epidemic by far the largest outbreak of Ebola in human history 4, this zoonotic event still does not even come close to the annual impact of pneumonia and influenza (3.5 million deaths in 2000) or tuberculosis (1.5 million deaths), diseases that have established themselves in human populations by optimizing their virulence level in human hosts.14 Just because the risk of filovirus adaptation to the human species is currently minimal, however, does not mean we should ignore the potential threat. As our ecological landscape changes due to a wide range of phenomena such as climate change and deforestation, which effectively expand the human-wildlife interface, the currently increasing frequency of zoonotic events is likely to continue and possibly even accelerate. As of this moment, we lack the tools to predict seemingly erratic Ebola and Marburg outbreaks. Most people agree that the most important defense against zoonoses is effective surveillance.9 Yet, as illustrated by the West African Ebola outbreak, the regions most at risk of these zoonotic events also lack effective public health surveillance.
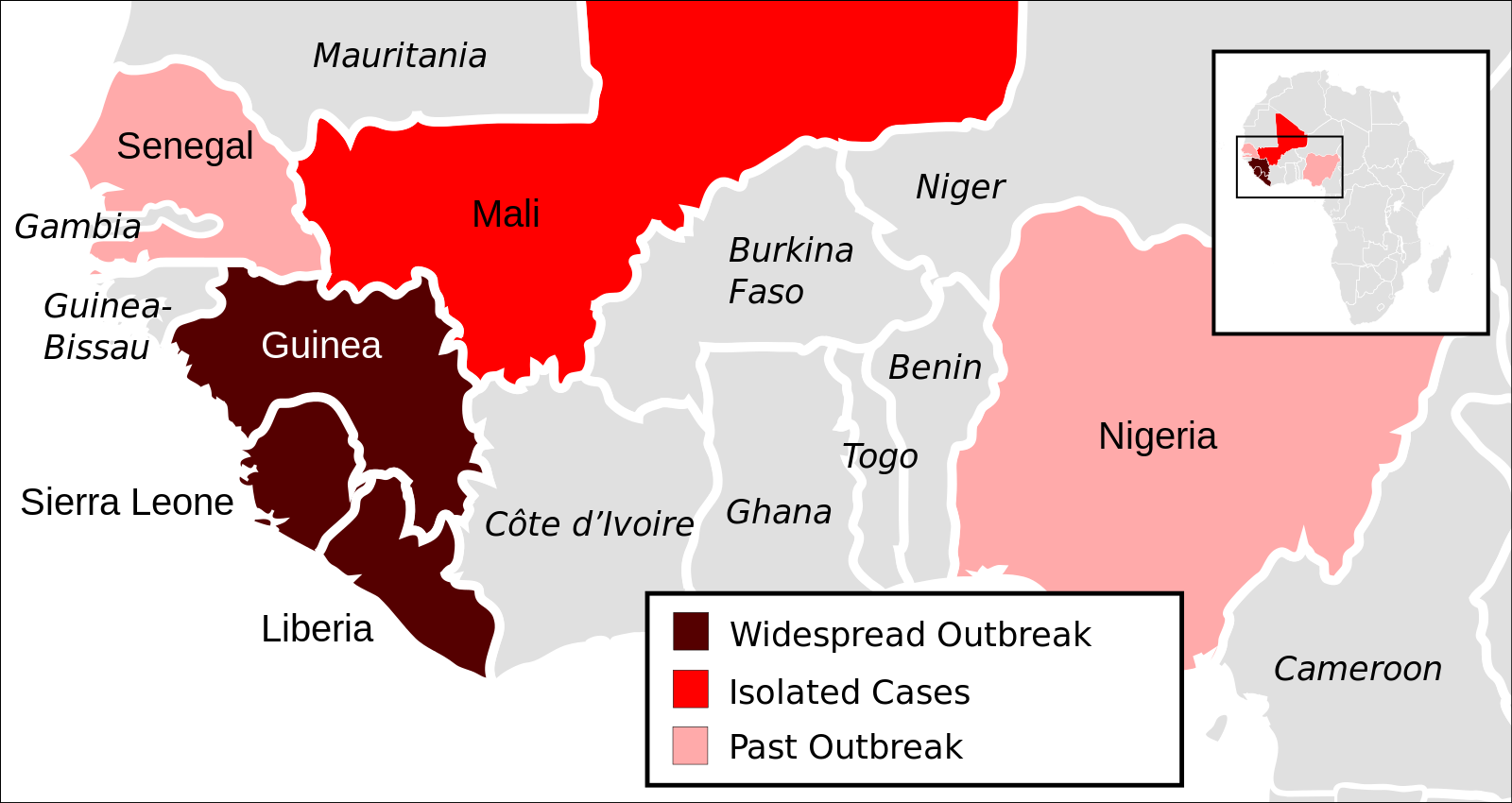
For decades, diseases common to sub-Saharan Africa and Southeast Asia were known as “tropical diseases;” in other words, diseases that did not typically affect those living in North America or Europe. But given the level of globalization today, an outbreak concerns regions beyond those in which it occurs. Viruses such as Ebola transmit rapidly, and have the potential to infect individuals around the globe within a few days or weeks. Significant attention has been given to the idea of an Ebola vaccine, with only limited success. Less attention has been paid to the importance of surveillance methods. Zoonotic diseases are particularly difficult to deal with from a public health perspective as they can retreat into their animal hosts until conditions line up that again facilitate spillover. Without comprehensive surveillance and contact tracing resources available in at-risk areas, we run the risk of seeing more frequent zoonotic events reminiscent of the 2014-2015 West Africa Ebola outbreak.
That is not to say, however, that virus hunting poses the solution. As noted by Professor Andrew P. Dobson of Princeton University, “Undiscovered viruses are abundant everywhere …and we need to think more about the dynamics of emergence than the romance of fishing for viruses with computer chips.”15 I would take that statement a step further: not only should we focus on the dynamics of emergence, but we also need to create infrastructural improvements that can improve the quality of daily life and strengthen disease response efforts in the event of an outbreak. One of the main problems with the West African Ebola epidemic response that many overlook is the fact that large numbers of cases occurred in communities with poor transportation and communication networks.16 Much of the focus in stopping Ebola centered around the creation of a vaccine, but what good can a vaccine do if the affected region lacks the infrastructure to distribute that vaccine to those in need? Similarly, how can we expect to survey “hotspot” regions when inadequate road and communication networks prevent at-risk communities from receiving the support that they need? What happens when an infected individual travels multiple days to a larger village or city to seek treatment and infects those he or she comes in contact with along the way? Discovering an outbreak represents an important first step. Developing a vaccine for filoviruses would be another major breakthrough. But adequate infrastructure to make the distribution of treatment and information possible, a factor all too frequently overlooked, must precede the implementation of any sort of surveillance system or vaccine distribution.

Zoonoses constitute a major public health risk already, and threaten to grow even more dangerous over the coming decades. There is no simple way to explain the phenomenon of spillover virulence without delving into the evolution of the virus and its hosts, as well as the intersection of humans, wildlife, and the environment. At this intersection, the human-wildlife-environment interface, the dreaded “species jump” occurs. But we must recognize that this species jump represents only the first step; the biology of the host and pathogen play an equally important role in determining how the virus will interact with its new target population. Filoviruses provide ample opportunity for research, particularly with regards to the role of bats as the natural reservoir of these pathogens. Spillover events are an inevitable consequence of living in a world where humans, wildlife, and pathogens all constantly evolve. We can, however, potentially slow the increase in frequency of spillover events and minimize the devastation they cause to the populations in which they emerge. Yet unfortunately that is not the future we are heading towards if we continue on our current trajectory.
______________
Rachel is a senior Global Affairs major in Branford College. She is also a Global Health Fellow and is participating in the 5 year BA/MPH program. She can be contacted at rachel.arnesen@yale.edu.
References
- Kreuder Johnson, C., et al (2015). Spillover and pandemic properties of zoonotic viruses with high host plasticity. Nature: Scientific Reports, 5(14830), 1-8.
- Burnet, F.M. & White, D. Natural History of Infectious Disease. Cambridge University Press, Cambridge, 1972.
- Croser, E.L. & Marsh G.A (2013). The changing face of the Henipaviruses. Veterinary Microbiology, 167(1-2), 151-158.
- Outbreaks Chronology: Ebola Virus Disease. (2015, November 12). Retrieved from http://www.cdc.gov/vhf/ebola/outbreaks/history/chronology.html.
- Roddy, P., et al (2010). Factors Associated with Marburg Hemorrhagic Fever: Analysis of Patient Data from Uige, Angola. Journal of Infectious Disease, 201(12), 1909-1918.
- Middle East respiratory syndrome coronavirus (MERS-CoV). (2014). Retrieved from http://www.who.int/emergencies/mers-cov/en/.
- O’Shea, T.J., et al (2014). Bat flight and zoonotic viruses. Emerging Infectious Diseases, 20(5), 741-745.
- Ebert, D., & Bull, J. (2008). The evolution and expression of virulence. In S. Stearns & J. Koella (Eds.), Evolution in Health and Disease (2nd ed., pp. 153-167). New York: Oxford University Press.
- Gortazar, C., et al (2014). Crossing the interspecies barrier: opening the door to zoonotic pathogens. PLoS Pathogens, 10(6), e1004129.
- Gagneux, S (2012). Host-pathogen coevolution in human tuberculosis. The Royal Society Publishing: Philosophical Transactions B, 367(1590), 850-859.
- Mills, J.N., Gage, K.L, & Khan, A.S (2010). Potential influence of climate change on vector-borne and zoonotic diseases: A review and proposed research plan. Environmental Health Perspectives, 118(11), 1507-1514.
- Peterson, A., Bauer, J.T., & Mills, J.N (2004). Ecologic and geographic distribution of filovirus disease. Emerging Infectious Diseases, 10(1), 40-47.
- Fa, J.E., Peres, C.A. & Meeuwig, J (2002). Bushmeat exploitation in tropical forests: an intercontinental comparison. Conservation Biology, 16(1), 232-237.
- World Health Organization (WHO) (1999). Report of the meeting of the Ad Hoc Committee on orthopoxvirus infections, 14-15 January, Geneva. Document No. WHO/CDS/CSR/APH/99.5. WHO, Geneva, 9 pp.
- Institute of Medicine of the National Academies (2014). The Influence of Global Environmental Change on Infectious Disease Dynamics: Workshop Summary. The National Academies Press: Washington D.C.
- CDC (2014, December 19). Challenges in Responding to the Ebola Epidemic – Four Rural Counties, Liberia, August-November 2014. Morbidity and Mortality Weekly Report, 63(50), 1202-1204.
