BY ISLA HUTCHINSON MADDOX
HIV/AIDS and Viral Load Monitoring in Uganda
At the 21st International AIDS Conference in Durban, South Africa in July 2016, a prevailing sense of optimism filled the room as thousands of the brightest minds in HIV/AIDS research and care flooded the Durban International Convention Centre. This annual conference provides a unique forum to discuss recent scientific developments, to examine necessary policy change, and to collectively reflect on differing perspectives on HIV across the globe.1 This year’s conference culminated in a rallying cry to adopt the UNAIDS “Fast-Track” strategy, which seeks to end the deadly epidemic by the year 2030.2 Many, however, fear that the convention only served to sensationalize progress and distract from many unaddressed social and economic barriers to care. “It is clear that the global rhetoric around the current ‘end of AIDS’ needs to be matched with an equally robust reality check,” wrote scientists Chris Beyrer, Linda-Gail Bekker, and Françoise Barrè-Sinoussi in Spotlight, a South African publication that monitors the nation’s TB and HIV response.3 My own “reality check” came this summer when I accepted an internship to assess the role of viral load monitoring at the Infectious Diseases Institute (IDI) in Kampala, Uganda.

Uganda, “the pearl of Africa,” is a landlocked East African country with a population of over 37 million people. In the 1990s, HIV/AIDS rates decreased dramatically in Uganda when Yoweri Museveni, the longstanding president, adopted a multi-sectoral approach that emphasized HIV/AIDS education and public awareness campaigns. In recent years, however, there has been an uptick in the number of infections, which many attribute to government complacency after this initial success.4 In 2015, UNAIDS reported that HIV prevalence rates for adults aged 15 to 49 stood at about 7.1%, with the majority of these infections clustered among at risk populations, such as young adults, commercial sex workers, men who have sex with men, and discordant couples (one partner is HIV+ and the other is HIV-).5 This departure from a “Cinderella success story” has brought renewed energy, funds, and focus to the country’s greatest health burden.5 A primary objective in Uganda since the renewal of PEPFAR (President’s Emergency Plan for AIDS Relief) in 2008 has been to expand access to treatment beyond the 57% of HIV+ adults who now receive some form of antiretroviral therapy (ART).5 Although it is essential to tackle the remaining 43%, it is equally important to ensure that hospitals and clinics deliver the best quality patient management when HIV+ adults do receive ART.6 In regards to this secondary goal, the strength of the HIV treatment cascade, a model that “outlines the sequential steps or stages of HIV medical care from initial diagnosis to achieving the goal of viral suppression,” becomes an especially important measurement of optimal medical care.7
There are many interventions along this cascade of care that aim to evaluate and to reinforce the progress that a patient makes during his or her path to viral suppression. The World Health Organization (WHO) defines viral suppression as less than 1000 copies of the HIV RNA virus per milliliter of blood.8 Many clinics invest in both CD4 counts9 and viral load (VL) monitoring as preliminary indicators of treatment failure or suboptimal adherence. Some researchers, however, herald VL monitoring as a wiser investment since it is more effective at detecting subtleties in immune response.9 For example, research has shown that people with the same CD4 cell counts may have different viral loads, and those with a higher VL are more likely to develop symptoms of an opportunistic infection than those with a lower VL.9 VL monitoring, therefore, has taken center stage in recent years in patient management: preventing unnecessary switches to second-line therapy and ensuring that resistance testing, adherence counseling, and VL rechecks remain crucial interventions along the cascade of care.10-11 VL testing is like an alarm that alerts physicians that the current drug regimen is no longer working for a patient. The course of HIV drugs may be simply rendered clinically ineffective or the patient may not be adhering properly to his or her daily cocktail of first-line drugs: AZT, a nucleoside reverse transcriptase inhibitor, and a non-nucleoside reverse transcriptase inhibitor.12 If adherence is suboptimal, this may allow for the accumulation of deleterious mutations among the proliferating HIV virion cells and subsequently lead to a drug resistant strain of infection. VL monitoring, therefore, forces the physician to focus on and better tailor care for those patients who are unable to achieve viral suppression, exhausting all possible interventions before switching a patient to a second-line course of therapy that is not only more toxic but also more expensive.13
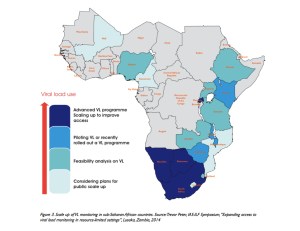
In 2014, UNAIDS created a stepping-stone along the path to eradication with the 90-90-90 global agenda: 90% diagnosed, 90% on treatment, 90% virally suppressed.14 At the International AIDS Conference, there was an overall consensus that global gains indicate a gradual progression towards this target; however, many sub-Saharan African countries have criticized the feasibility of this goal recently with the deadline fast approaching in just four years.15 Viral load monitoring, a central component of the viral suppression portion of the 90-90-90 goal, is an especially resource-intensive practice that is of greatest consequence in the most resource-deprived settings across sub-Saharan Africa. Only a few nations, including South Africa, Botswana, and Namibia have scaled up VL programs across the country, while Uganda is still in a phase of data collection and continues to conduct feasibility analysis in both urban and rural parts of the country (Figure 1).16 This analysis or research raises a myriad of questions. Does VL monitoring realistically and efficiently fit into Uganda’s cascade of care? For those clinics where it is currently in use, are physicians properly utilizing it? Do certain trends emerge along this cascade of care that might bring to light gaps in patient management? Implementation research, therefore, becomes essential to answering these questions and ultimately determining whether this resource-intensive practice is worth the investment.
Uganda has, therefore, shifted research priorities away from the assessment of new medical technologies and towards implementation research in order to determine whether the technologies currently in play are effective. Implementation research is a relatively new field of health research that evaluates the practice of medicine – not from the confines of a sterile, well-controlled laboratory – but in real world conditions.21 Public health experts and scientists can work together to not only build new and more refined technologies but also to ensure that they will be properly utilized “on the ground.” Uganda is in the stages of assessing the quality of VL monitoring programs currently in place at some of the nation’s largest clinics in order to determine if one might serve as the most efficient and practical model of care in this developing country. The Infectious Disease Institute (IDI) at the heart of Uganda’s capital, Kampala, regularly serves 8000 HIV infected patients and therefore provided the perfect backdrop to conduct our research this summer.
Viral Load Monitoring in Uganda: The Basics of our Study
Previous studies conducted in 2015 at IDI had already examined the importance of VL monitoring as a litmus test or measurement of adherence to treatment among the clinic’s general population. As a result, we refined our research to analyze the care of young adults, aged 18-25.
Globally, young adults have been identified as an especially vulnerable population.22 Sub-Saharan Africa alone accounts for 70% of the world’s new HIV infections among young people aged 15-24.22 They are more likely to be inexperienced with regards to strenuous ART drug regimens, engage in risky sexual behaviors, and to have higher rates of suboptimal adherence compared to older HIV patients.19 A meta-analysis conducted in 2013 reviewed published literature on HIV among adolescents and young adults (AYA) and found that only 62% were adherent to ART drug regimens, far below number needed to achieve the third 90-90-90 target.20 An internal assessment by the Quality Assurance Office at the Infectious Disease Institute had found that about 84% of the general patient population (N = 6438) had achieved viral suppression over the course of about a single year, therefore, we used this as our benchmark – initially hypothesizing that the young adult patient body would present much lower numbers.
Our team conducted a retrospective cohort study of young adult patients undergoing routine VL monitoring between December 2014 to July 2016. As per World Health Organization guidelines, a VL test is conducted every 6-12 months for all patients on ART.21-22 To assess the effectiveness of IDI’s VL monitoring protocol, data were extracted from a clinician’s “decision form” that is filled out for those patients with a detectable viral load (>1000 copies/mL). This clinician form lists a number of possible directions for patient care that essentially builds IDI’s stepwise protocol for a detectable patient, similar to an HIV treatment cascade. Some physicians, for example, immediately refer patients to adherence counseling after a detectable viral load, reflecting suspected suboptimal adherence. Other patient charts are referred to a switch meeting: a multidisciplinary meeting unique to IDI, attended by counselors, nurses, pharmacists, and clinicians each week. During these hour-long switch meetings, counselors present the case history of patients who have been unable to achieve viral suppression. The team then makes a comprehensive decision regarding the status and care of a single patient. We calculated the median turnaround time for various interventions along the cascade (e.g. the time between detectable viral load and switch to second-line) in order to determine how well IDI managed patient care from day to day. Turnaround time is an especially important measurement along this treatment cascade as a number of patients may fall through the cracks with little to no safeguards in place to get them back on track and returning regularly at the clinic.
Between December 2014 and July 2016, 252 young adult patients were tested for VL at IDI, of which 48 (19%) had a VL >1,000 copies/mL (viral load detectable). 370 total VL tests were performed, of which 81% (300) were undetectable. Figure 2 summarizes the actions taken for those 48 patients who collectively had 70 detectable VLs over the course of the study with green boxes highlighting important trends.
Of 48 patients with a detectable VL, only 21 switched to second-line treatment (43.8%), with one patient switching twice over the course of the study. In an attempt to prevent unnecessary switches, 10 of these 22 switches (45.5%) occurred only after adherence counseling and a VL recheck. Eleven switches (50.0%) occurred following a switch meeting. Of those 21 patients who switched, 16 attended at least one adherence counseling session following the switch in order to highlight the necessity for proper adherence. Four patients (50.0%) who received a resistance profile switched to a second-line regimen, often prior to receiving resistance results. The median time to switch from the date of the initial VL test was 146 days (IQR: 91-209).
Throughout the course of the study, 22 patients (8.7%) qualified as lost to program (LTP), signifying that their last clinic visit was more than 3 months before the study’s termination. A majority of these LTP patients however had an initial undetectable VL (n=14, 63.6%), while 8 (36.4%) had an initial detectable VL. Three patients (1.2%) were unable to be tracked, 2 patients (2.4%) died, and 4 patients (4.8%) transferred out of IDI during the study period. At the end of the study, 21 (43.8%) of the original 48 patients with a detectable VL remain on first-line ART and continue to attend IDI regularly.
Discussion
While the third 90-90-90 target set by UNAIDS provides a lofty goal to strive towards with regards to patient care and VL monitoring, this study found that young adults, an at-risk population, came close to this target with over 81% of patients achieving viral suppression. As clinics strive to close this gap, it is essential that those clinics similar to IDI apply additional safeguards when treating patients. In order to ensure that this target is feasible for all populations, certain protective measures should be put in place, along with the cascade of care, that take into account potential risk factors that might impede optimal adherence. Qualitative research, therefore, should be incorporated into future studies in order to better assess what circumstances or factors might cause to a patient to drop out of treatment or to adhere improperly to their drug regimen. Variables might include sex, age, education, religion, number of current sexual partners, and marital status.
Delaying switches to second-line therapy appears especially important for young adult patients who are often ART naïve, adjusting to a daily drug regimen, and facing internal and external stigma. During this study, less than half of patients with a detectable VL switched to second-line treatment, suggesting clinician’s suspicion of suboptimal adherence. For these detectable patients, adherence counseling may play a crucial role in preventing unnecessary switches to second line therapy and helping patients achieve viral suppression. Complementary interventions and a stringent stepwise protocol that recognizes the potential leaks in the cascade of care are especially important for this transient, vulnerable population.
One component along this cascade of care that was called into question during this study was the role of resistance testing in patient management. Blood samples for resistance profiles are often unable to be analyzed at an in-house location and are sent as far as the United States. Consequently, the turnaround time is great. Patients often switch to second-line prior to receiving the resistance profile or become lost to program. The young adult population is more likely to exhibit suboptimal adherence and therefore accumulate resistant mutations, and data suggest that physicians switch these patients regardless of resistance testing results.23 Resistance testing therefore may not be the most decisive intervention in a resource-low setting especially when routine VL monitoring is so resource-intensive and has proven to be a more practical investment.
Although this study is in part strengthened by the fact that it follows patients through the entire process of VL monitoring at a centralized location, a number of limitations arise when performing such condensed and streamlined research. Primarily, the generalizability of these findings may be limited. Uganda has a relatively young ART / VL monitoring program; therefore, these findings might hold little external validity when compared to naïve programs in other Sub-Saharan countries or when compared to more robust programs, like those in South Africa.24 The study’s cohort was limited to young adults who attend IDI; therefore, the sample size was small and only tracked for a period of 20 months.
This study evaluated the effectiveness of IDI’s protocol alone and did not use a control group to compare the strength of other VL monitoring protocols. Further research must be conducted to not only strengthen existing protocols but also to determine the best possible cascade of care when managing HIV+ patients. In addition, expanding access to ART for all HIV+ patients will ensure that more patients regardless of location can begin this cascade of care and achieve viral suppression. This research ultimately reflects the importance of complementary interventions such as adherence counseling and viral load rechecks when managing HIV+ patients especially within the young adult population. In order to ensure that the 90-90-90 target is met in the next four years, it is essential to continue research at IDI.
Final Reflections on HIV/AIDS and Uganda
This summer, I was forced to not only confront my conception of HIV/AIDS – a disease that I have studied furiously in the classroom, reading countless policy briefs and UNAIDS reports – but also to address my conception of Uganda. While working at an HIV clinic, it was unnerving at times to hear how casually physicians and counselors spoke about this horrifying disease that I was witnessing for the first time. I saw people on the brink of death – severely malnourished as their diaphragms and lungs painfully ached to breathe. The doctors, on the other hand, saw patients. This is not to minimize how the physicians approached each person in need; however, they as a community needed some form of humor in order to continuously provide the critical care that they do. There is obviously stigma surrounding HIV in Uganda; however, here at IDI it is not apparent. This is their job, and this is their reality. When I traveled to Africa for the first time, my understanding of the continent was the one that is plastered on billboards and pictured in advertisements for NGOs, UNICEF, and Save the Children. Living in Uganda, however, has allowed me to step outside of the white-washing treatment and homogenization that the Western media gives this continent as a whole. I had all these preconceived notions about what Africa would be like. Yes there is extreme poverty, hunger, and rampant disease; and there are as many posters to remind you to wear a condom as there are sushi joints in New Haven, but there are also millions of happy people – that love Uganda; that have – what we would call – normal lives. They go to bars and look forward to the movies on Monday nights and obsess over Facebook and Instagram likes. Although this article doesn’t explicitly serve as a plug for Yale International Coordinated Internships, I believe that my experience abroad this summer was invaluable. In recent years, global health as a discipline has gained ground as thousands of public health experts, scientists, and physicians flock to this field of work. While it is a remarkable feat, it is essential that studies remain as closely grounded to the field as possible. With this perspective and focus, it will serve as a constant reminder that these research subjects or patients are real people, not merely figures or statistics. It is one thing to study “HIV/AIDS in Africa” from the confines of the classroom, but another thing entirely to witness its toll on real people.
Isla Hutchinson Maddox is a sophomore in Saybrook College majoring in Applied Mathematics. She can be contacted at grace.kang@yale.edu.
__________
References:
- AIDS Abstract Book: 21st International AIDS Conference. (2016). Retrieved from http://www.aids2016.org/.
- UNAIDS. (2014). Fast-Track: Ending the AIDS Epidemic by 2030. Retrieved from http://www.unaids.org/en/resources/documents/2014/JC2686_WAD2014report.
- Beyrer, C., Bekker, L.G., and Françoise Barré-Sinoussi. (2016). It’s too soon to speak of the end of AIDS. Retrieved from http://www.spotlightnsp.co.za/2016/07/18/soon-speak-end-aids/.
- Kron, Josh. (2012). In Uganda, an AIDS Success Story Comes Undone. Retrieved from http://www.nytimes.com/2012/08/03/world/africa/in-uganda-an-aids-success-story-comes-undone.html?_r=0.
- UNAIDS. (n.d.) Uganda Country Profile. Retrieved from http://www.unaids.org/en/regionscountries/countries/uganda.
- UNAIDS. (2016). Prevention Gap Report. Retrieved from http://www.unaids.org/sites/default/files/media_asset/2016-prevention-gap-report_en.pdf.
- HIV Continuum of Care. (2016). Retrieved from https://www.aids.gov/federal-resources/policies/care-continuum/.
- World Health Organization. (2014). Technical and Operational Considerations for Implementing HIV Viral Load Testing.
- Viral Load: Looking at CD4 and viral load together. (2016). Retrieved from: http://www.aidsmap.com/Viral-load/page/1327496/#item1327515.
- Kantor R, Diero L, Delong A, et al. (2009). Misclassification of first-line antiretroviral treatment failure based on immunological monitoring of HIV infection in resource-limited settings. Clin Infect Dis, 29: 454-62.
- World Health Organization. (2015). Consolidated Guidelines on the Use of Antiretroviral Drugs for Treating and Preventing HIV Infection.
- World Health Organization. (2014). Technical and Operational Considerations for Implementing HIV Viral Load Testing.
- Greig J, du Cros P, Klarkowski D, et al. (2011). Viral load testing in resource-limited setting: quality control is critical. Journal of the International AIDS Society, 14: 23.
- UNAIDS. (2014). 90–90–90: an ambitious treatment target to help end the AIDS epidemic. Retrieved from http://www.unaids.org/sites/default/files/media_asset/90-90-90_en_0.pdf.
- UNICEF. (2011). Opportunities in Crisis: Preventing HIV From Early Adolescence to Young Adulthood. Retrieved from http://www.unicef.org/publications/index_58708.html.
- Medecins Sans Frontieres. (2014). Achieving Undetectable: What Questions Remain in Scaling-Up HIV Virologic Treatment Monitoring? Retrieved from https://www.msfaccess.org/sites/default/files/MSF_IssueBrief_undetectable6.pdf.
- Peters, D., Taghreed, Alonge, Olakunle, et al. (2013). Implementation research: what it is and how to do it. British Medical Journal 347: f6753.
- Global Report: UNAIDS report on the global AIDS epidemic 2013.
- UNICEF. (2011). Opportunities in Crisis: Preventing HIV From Early Adolescence to Young Adulthood. Retrieved from http://www.unicef.org/publications/index_58708.html.
- Adherence to antiretroviral therapy in adolescents living with HIV: systematic review and meta-analysis.
- World Health Organization. (2014). Technical and Operational Considerations for Implementing HIV Viral Load Testing.
- Baggaley R., et al. (2015). Young key populations and HIV: a special emphasis and consideration in the new WHO Consolidated Guidelines on HIV Prevention, Diagnosis, Treatment, and Care for Key Populations. Journal of the International AIDS Society 18:1.
- Charles, M, Noel F, Leger P, et al. (2008). Survival, plasma HIV-1 RNA concentrations and drug resistance in HIV-1-infected Haitian adolescents and young adults on antiretrovirals. Bulletin of the World Health Organization 86: 970-977.
- Stevens, Wendy and TM Marshall. (2010). Challenges in Implementing HIV Load Testing in South Africa. Journal of the International AIDS Society 201: S78-S84.

